Search results
Search for "silver(I) catalyst" in Full Text gives 2 result(s) in Beilstein Journal of Organic Chemistry.
Self-assembled coordination thioether silver(I) macrocyclic complexes for homogeneous catalysis
Beilstein J. Org. Chem. 2019, 15, 2465–2472, doi:10.3762/bjoc.15.239
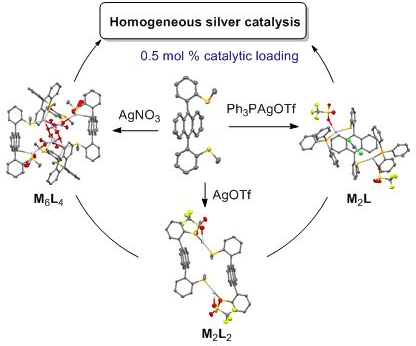
- slowly added to afford a precipitation. After filtration, the isolated solid was washed with diethyl ether and dried under vacuum. General procedure for the acetalization/cycloisomerization of alkyne 2: An oven-dried Schlenk tube was charged with the silver(I) catalyst (0.5–5 mol %), then degassed and
Synthesis of 2H-furo[2,3-c]pyrazole ring systems through silver(I) ion-mediated ring-closure reaction
Beilstein J. Org. Chem. 2019, 15, 679–684, doi:10.3762/bjoc.15.62
- cyclization of readily available 4-alkynyl-3-hydroxy-1H-pyrazoles can be used as an efficient method to access many novel 2,5-disubstituted 2H-furo[2,3-c]pyrazoles. Keywords: 5-endo-dig cyclization; 2H-furo[2,3-c]pyrazole; pyrazole; silver(I) catalyst; Sonogashira coupling; Introduction Heterocyclic ring
- synthetic methods has been developed for the construction of carbon–heteroatom bonds using these types of catalysts. For example, the gold(I) catalyst [(Ph3PAu)3O]BF4 was applied in the regioselective intramolecular cyclization of alkynols to construct bicyclic ethers [37], while the silver(I) catalyst
- -pyrazoles. The desired 5-endo-dig cyclization leading to the formation of the 2H-furo[2,3-c]pyrazole ring system is easily achieved by heating the aforementioned hydroxyalkynyl substrates with a base in DMF in the presence of a silver(I) catalyst. a) ORTEP diagram of the asymmetric unit consisting of two
















